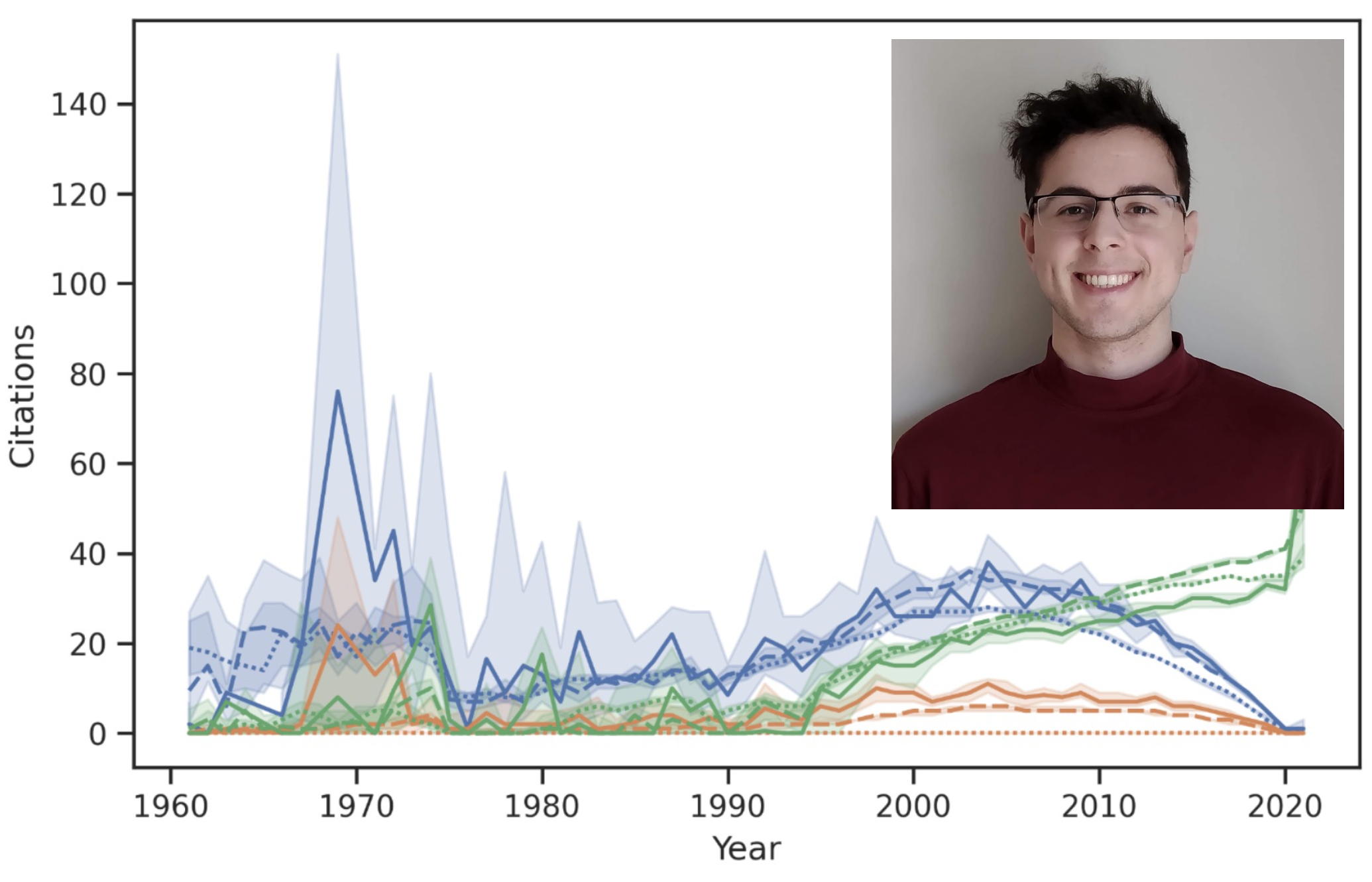
Keaton W Smith, Brian P Alcock, Shawn French, Maya A Farha, Amogelang R Raphenya, Eric D Brown, & Andrew G McArthur. Microbiol Spectrum. 2023 Nov
Category: antibiotic resistance

IIDR Trainee Day 2023
COLIN BRUCE – Investigating Fibre Degradation in the Infant Gut Microbiota ; DIRK HACKENBERGER – Was World War 2 Foundational to the Antimicrobial Resistance Crisis?

The McArthur lab welcomes Brody Duncan, M.D. (Hamilton Health Sciences) as he starts his M.Sc. studies with us, investigating standards and methods for clinical reporting

Baker, S.J.C., J. Maciejewski, M.-T. Usuanlele, J. Gilchrist, D.R. Sharma, D. Bulir, M. Smieja, M. Loeb, M.G. Surette, A.G. McArthur, & D. Mertz. 2023. Investigating

Today the Comprehensive Antibiotic Resistance Database turns 10 years old! While it started a few years before this in the laboratory of Dr. Gerry Wright,

CARD and CZ ID are thrilled to launch a new CZ ID module that allows researchers to detect and analyze antimicrobial resistance (AMR) genes in
Volunteer starting in 2019, thesis student, summer student, Masters student (2020-2022), and developer, Arman Edalatmand has been a great lab member for over 4 years,

Alcock, B.P., A.R. Raphenya, A. Edalatmand, & A.G. McArthur. 2023. The Comprehensive Antibiotic Resistance Database – curating the global resistome. Oral presentation at the 16th

Arman Edalatmand & Andrew G McArthur. 2023. Database, doi: 10.1093/database/baad023. Scientific literature is published at a rate that makes manual data extraction a highly time-consuming
CBW & VTEC 2023
They’re back! Thanks to Karyn Mukiri, Jalees Nasir, and Madeline McCarthy for serving as Teaching Assistants for the re-booted Canadian Bioinformatics Workshop in Infectious Disease

The McArthur Lab welcomes Team Meta-Model, a volunteer curation team led by former BioPharm Co-Op Keaton Smith, working on curating and upgrading the Comprehensive Antibiotic

IIDR Trainee Day 2022!
KARYN MUKIRI – Predicting the total resistome KEATON SMITH – Advancements in curation of the Comprehensive Antibiotic Resistance Database JALEES NASIR – Viral fishing expedition:

Alcock et al. 2023. Nucleic Acids Res Oct 20, 2022:gkac920. (online ahead of print) The Comprehensive Antibiotic Resistance Database (CARD; card.mcmaster.ca) combines the Antibiotic Resistance

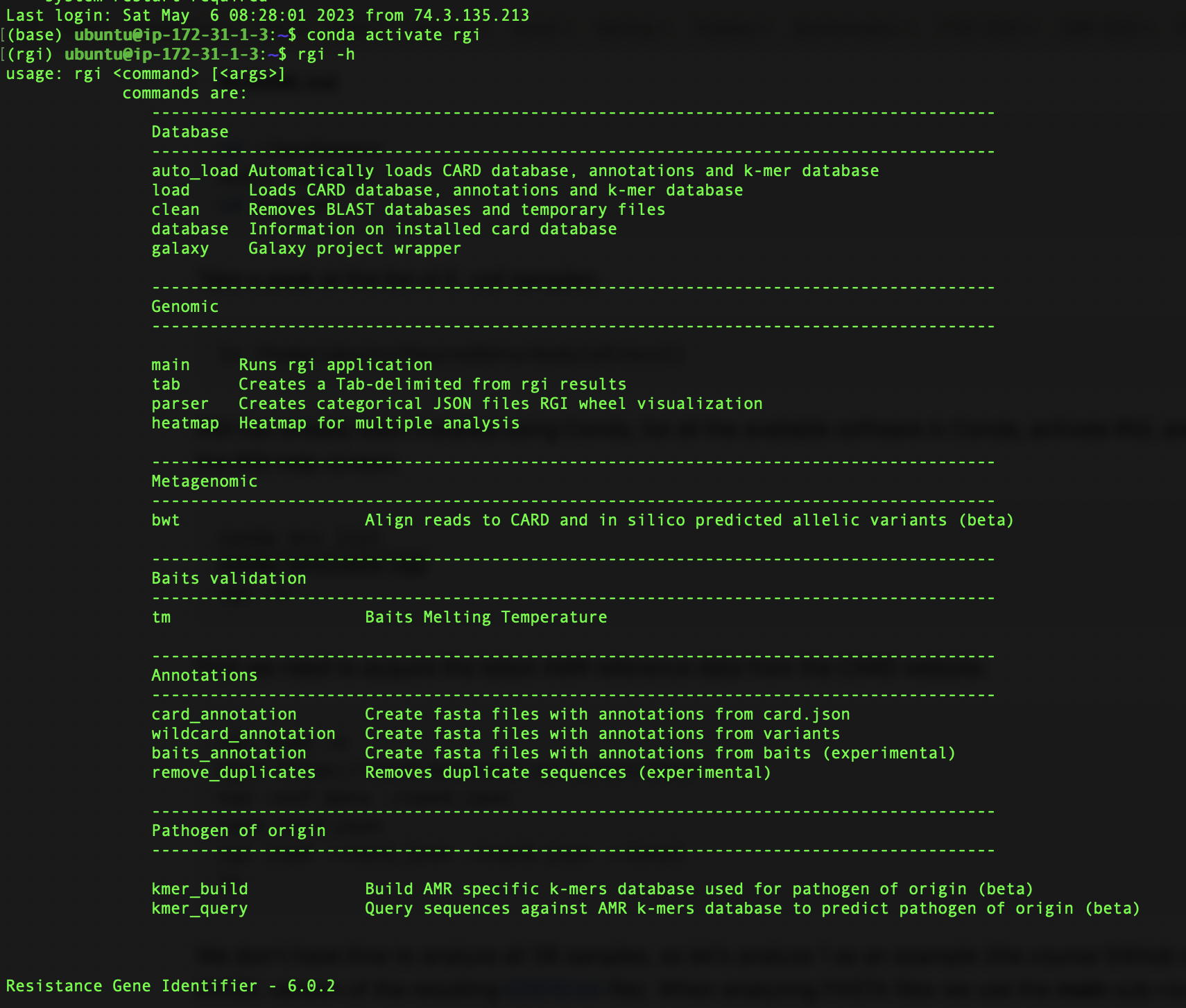
RGI version 6.0.0 – Major release, with parameter and reference data loading changes that may impact scripts. Please see updated README. Added an option (–keep)

The McArthur lab welcomes back past undergraduate student and now newly minted Biochemistry & Biomedical Sciences M.Sc. student Mateusz Wlodarski! Mateusz will be working on

Guitor et al. 2022. Microbiome 10(1):136. Background: Probiotic use in preterm infants can mitigate the impact of antibiotic exposure and reduce rates of certain illnesses;

Summer Conferences 2022
(presenters in bold; lab members underlined) Baker, S.J.C., S. Yaqoob, M.-T. Usuanlele, J. Gilchrist, M. Smieja, M. Loeb, S. Khan, J. Pernica, M.G. Surette, A.G.

Raphenya et al. Scientific Data volume 9, Article number: 341 (2022). Whole genome sequencing (WGS) is a key tool in identifying and characterising disease-associated bacteria

The burden of drug-resistant infections continues to threaten our arsenal of antibiotics. Despite the rise in genomic sequencing of clinical isolates, there is a lack

